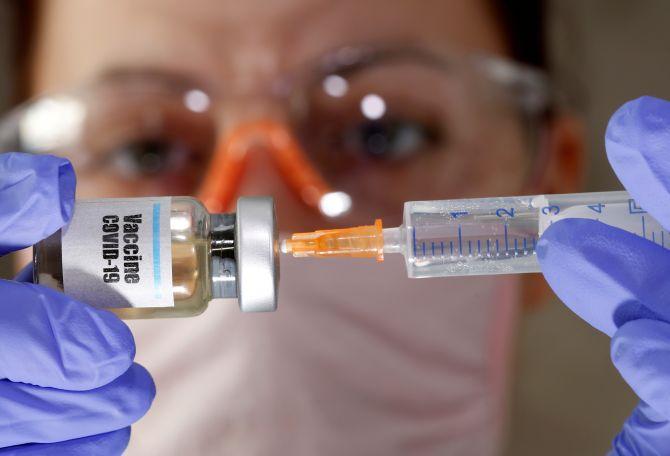
The Drugs Controller General of India has given a nod to Serum Institute of India for conducting phase 2 and 3 human clinical trials of the Oxford University's COVID-19 vaccine candidate in the country, according to the health ministry.

Government officials said that DCGI Dr V G Somani granted the approval late Sunday night after a thorough evaluation based on the recommendations of the Subject Expert Committee on COVID-19.
"The firm has to submit safety data, evaluated by the Data Safety Monitoring Board, to the Central Drugs Standard Control Organisation before proceeding to phase 3 clinical trials," a senior official said, referring to the CDSCO.
"As per the study design, each subject will be administered two doses four weeks apart (first dose on day one and second dose on day 29) following which the safety and immunogenicity will be assessed at predefined intervals," the official said.
The DCGI has given approval to SII, Pune, to conduct Phase II+III clinical trials of Oxford University-Astra Zeneca COVID-19 vaccine (COVISHIELD) in India and this will hasten the development of the COVID-19 vaccine, the health ministry said in a statement.
As a rapid regulatory response, the expert panel at the CDSCO on Friday, after a detailed deliberation and considering the data generated on the vaccine candidate in phase 1 and 2 of the Oxford University trial, had recommended granting permission for phase 2 and 3 clinical trials of the potential vaccine, 'Covishield', on healthy adults in India, the officials also said.
Currently, phase 2 and 3 clinical trials of the Oxford vaccine candidate is going on in the United Kingdom, phase 3 clinical trial in Brazil and phase 1 and 2 clinical trials in South Africa.
The officials also said the SII had submitted a revised proposal on Wednesday after the SEC on July 28, following deliberation over its application, asked it to revise its protocol for the phase 2 and 3 clinical trials besides seeking some additional information.
The panel had also recommended that the clinical trial sites which have been proposed for the study be distributed across India.
According to the revised proposal by the SII, 1,600 people aged above 18 years will participate in the trials across 17 selected sites, including AIIMS-Delhi, B J Medical College in Pune, Rajendra Memorial Research Institute of Medical Sciences in Patna, Post Graduate Institute of Medical Education and Research in Chandigarh, AIIMS-Jodhpur, Nehru Hospital in Gorakhpur, Andhra Medical College in Visakhapatnam and JSS Academy of Higher Education and Research in Mysore.
"According to the application, it would conduct an observer-blind, randomised controlled study to determine the safety and immunogenicity of 'Covishield' on healthy Indian adults," the official said.
The SII, which has partnered with AstraZeneca, for manufacturing the Oxford vaccine candidate for COVID-19, had submitted its first application to the DCGI on July 25 seeking permission for conducting the phase 2 and 3 trials of the potential vaccine.
Initial results of the first two-phases of trials of the vaccine conducted in five trial sites in the UK showed that it has an acceptable safety profile and homologous boosting increased antibody response, sources had said.
To introduce the vaccine, SII, the world's largest vaccine maker by number of doses produced and sold, has signed an agreement to manufacture the potential vaccine developed by the Jenner Institute (Oxford University) in collaboration with British-Swedish pharma company AstraZeneca.