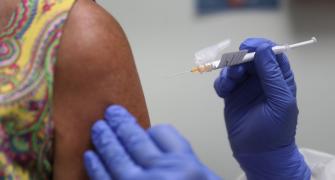
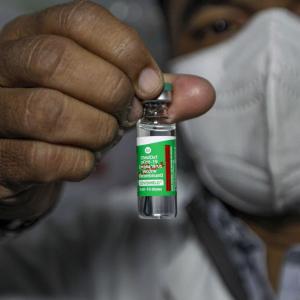
Twenty-five hospitalisations, which is 0.0005 per cent of the total people who received vaccine jabs, and 19 deaths not causally related to the vaccination presently have been reported till February 4, the Rajya Sabha was informed on Tuesday.
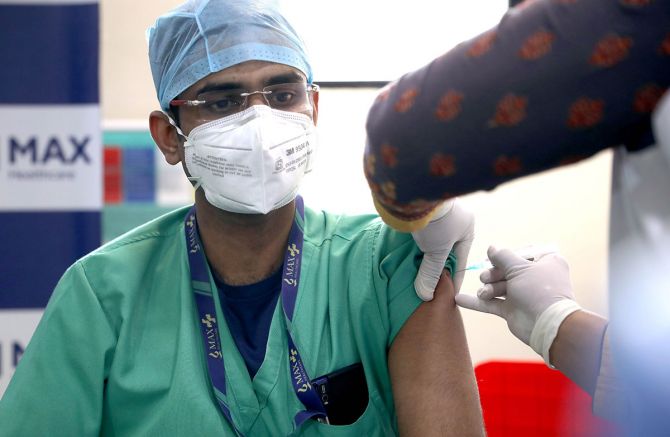
The nationwide COVID-19 vaccination drive began from January 16.
Two vaccines -- Covishield, manufactured by Serum Institute of India, and Covaxin by Bharat Biotech -- have been granted permission for restricted use in emergency situation by the national regulator, said Minister of State for Health Ashwini Choubey said in a written reply.
Asked whether there was any report of serious side effect after vaccination, he said, "A total of 25 hospitalizations i.e. 0.0005 per cent of total beneficiaries administered the first dose and 19 deaths i.e. 0.0004 per cent of total vaccinations till February 4, 2021, not causally related to vaccination presently, have also been reported."
On Adverse Effects Following Immunisation arising from the use of Covaxin and Covishield, Choubey said till February 4 total 81 AEFIs, which is 0.096 per cent of the total beneficiaries who have been administered Covaxin, have been reported.
For Covishield vaccine, the total number of AEFI stands at 8,402 which is 0.192 per cent of total beneficiaries vaccinated, the minister said.
Most of these are minor AEFIs like anxiety, vertigo, giddiness, dizziness, fever, pain, rashes, and headache which are self-limiting and all people have recovered.
According to the New Drugs and Clinical Trials Rules, 2019, under the Drugs and Cosmetics Act, 1940, and in light of urgent need due to coronavirus pandemic in the country, the Central Drugs Standard Control Organization headed by the Drug Controller General of India has permitted manufacture of two COVID-19 vaccines based on prescribed procedure and due evaluation of pre-clinical and clinical trial data.
The common side effects of vaccines under the country's immunization programme include pain, swelling, redness at injection site, local abscess, fever, malaise and others, Choubey said.
"The adverse events which have been reported from COVID-19 vaccines approved for restricted use in emergency situation include headache, rash, chills, myalgia, fatigue, fever, dizziness, inflammation and pain, swelling or redness at the site of injection, vaccination site, erythema, pruritus etc," he said.
So far, Bangladesh, Saudi Arabia, Morocco, Myanmar, Nepal, Afghanistan, Srilanka, Bhutan, the Maldives, Oman, Bahrain, Mauritius, Seychelles, Brazil, South Africa and Mongolia have finalised import of Indian COVID-19 vaccines, the minister informed the Upper House.
Choubey said AEFI cases are monitored through a well-structured and robust surveillance system that includes district, state and national-level committees.
On what actions have been taken by the government to overcome the hesitancy among the citizens to receive vaccines, the minister of state for health said COVID-19 Vaccines Communication Strategy has been prepared to raise awareness about the vaccines and address misinformation against immunisation.
The strategy aims at prompt dissemination of factual information about the vaccines and the vaccination process. This is being done through multimedia platforms (print/electronic/social media) and engagement of various stakeholders, community leaders and influencers to reach out to the last mile, he said.