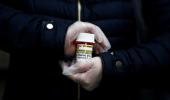
Hetero Healthcare on Wednesday said is set to deliver 20,000 vials of its antiviral drug Covifor (remdesivir) across the country for the treatment of COVID-19, at a maximum retail price of Rs 5,400 per vial.

The company is set to deliver the first set of 20,000 vials in two equal lots of 10,000 each, one of which will be immediately supplied to Hyderabad, Delhi, Gujarat, Tamil Nadu, Mumbai and other parts of Maharashtra, Hetero Healthcare said in a statement.
The other lot will be supplied to Kolkata, Indore, Bhopal, Lucknow, Patna, Bhubaneshwar, Ranchi, Vijayawada, Cochin, Trivandrum and Goa within a week, it added.
The company said it has fixed a maximum retail price of Rs 5,400 per vial for the drug.
On Tuesday, pharma major Cipla said, it will price its generic version of remdesivir at less than Rs 5,000 per vial.
It also said that the drug will be available in the next 8 to 10 days.
"The launch of Covifor in India is a significant milestone for all of us... Through Covifor, we hope to reduce the treatment time of a patient in a hospital, thereby reducing the increasing pressure on the medical infrastructure, overburdened currently due to accelerating COVID-19 cases," Hetero Healthcare MD M Srinivasa Reddy said.
The company is working closely with the government and medical community to make 'Covifor' quickly accessible to both public and private healthcare settings across the country, he added.
Covifor is the first generic brand of remdesivir, indicated for the treatment of COVID-19 patients in adults and children, hospitalised with severe symptoms of the disease, the statement said.
The drug is available in 100 mg vial (injectable). It needs to be administered intravenously in a hospital, critical care setting, under the supervision of a registered medical practitioner, it added.
On Sunday, Hetero said it has received the manufacturing and marketing approval for remdesivir from the Drug Controller General of India (DCGI) for the treatment of COVID-19.
In May, domestic pharma firms Hetero, Cipla and Jubilant Life Sciences entered into non-exclusive licensing agreements with drug major Gilead Sciences Inc for manufacturing and distribution of remdesivir.
The medicine has been issued an Emergency Use Authorization (EUA) tag by the United States Food and Drug Administration (USFDA) to treat COVID-19 patients.