The interim results of the World Health Organisation Solidarity Trial' indicate that four repurposed drugs for COVID-19 -- remdesivir, hydroxychloroquine, lopinavir/ritonavir and interferon regimens -- appeared to have little or no effect on reducing mortality, initiation of ventilation and duration of hospital stay.
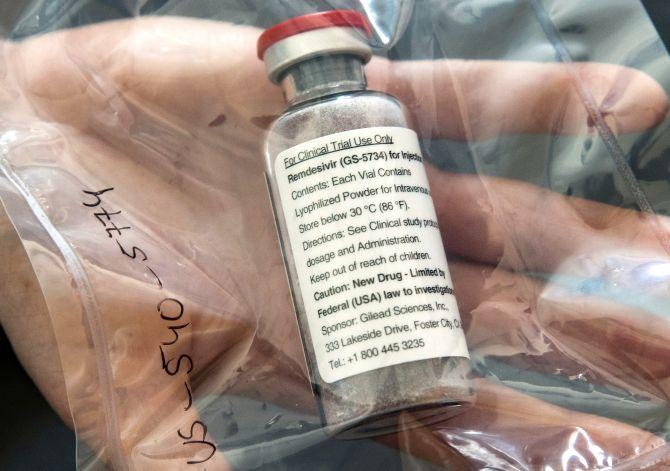
The interim findings of the trial in which the four drugs were tested in 405 hospitals across 30 countries, covering 11,266 adults, were released on 'medRxiv', a preprint server, on Thursday. The findings are under review for publication in a medical journal.
"In 405 hospitals across 30 countries, 11,266 adults were randomised, with 2,750 of them given remdesivir, 954 hydroxychloroquine, 1,411 lopinavir, 651 interferon plus lopinavir, 1,412 only interferon, and 4,088 no study drug," the uploaded trial study stated.
The trial arms related to hydroxychloroquine and lopinavir were discontinued earlier as the interim results had showed no benefit.
"The remdesivir, hydroxychloroquine, lopinavir and interferon regimens appeared to have little or no effect on hospitalised COVID-19 patients, as indicated by overall mortality, initiation of ventilation and
duration of hospital stay," the study said.
The findings hold significance as the use of remdesivir (for restricted emergency use purposes) has been recommended for treating coronavirus patients in moderate stage of the illness as "investigational
therapies" in Clinical Management Protocol for COVID-19 issued by the Union health ministry.
Other uses of the drugs, for example, in treatment of patients in the community or for prevention, would have to be examined using different trials, the WHO said.
The WHO Solidarity Trial was conducted from March 22 to October 4.
Meanwhile, the Indian Council of Medical Research on Friday said it was an active partner in the Solidarity Therapeutics Trial conducted under the aegis of the World Health Organization.
"The trial tested four repurposed drugs, namely, remdesivir, interferon 1a, lopinavir/ritonavir and hydroxychloroquine. Interim analysis showed that no benefits of remdesivir in any groups of COVID-19
(asymptomatic/mild/moderate/severe/critical) patients," it said in a statement.
The ICMR has succeeded in conducting this large randomised controlled study even during a pandemic situation and earlier lockdown. The study reliably answers critical public health questions concerning
therapeutics.
"Earlier, ICMR conducted PLACID trial for convalescent plasma indicating no benefit of it in COVID-19 treatment," the apex health research body said.
This was a well-coordinated national effort led by ICMR-National AIDS Research Institute, Pune with scientist Sheela Godbole at the helm, it said.
"The trial comprised 26 actively randomising sites with 937 participants in India. We are grateful to the trial participants and their families for contributing to these crucial findings," Samiran Panda, Head,
ICMR-Division of Epidemiology and Communicable Diseases, and director, ICMR-NARI said.
The Solidarity Therapeutics Trial is the world's largest global randomised controlled trial in a pandemic situation for COVID-19 therapeutics, spanning 30 countries. India contributed one-tenth of the participants in the trial, the statement added.