The elderly population above 60 years of age with comorbidities who are eligible for COVID-19 'precautionary doses' will have to take a medical certificate to take the dose, informed chief executive officer of National Health Authority Dr RS Sharma, who also heads the functioning of CoWIN platform.
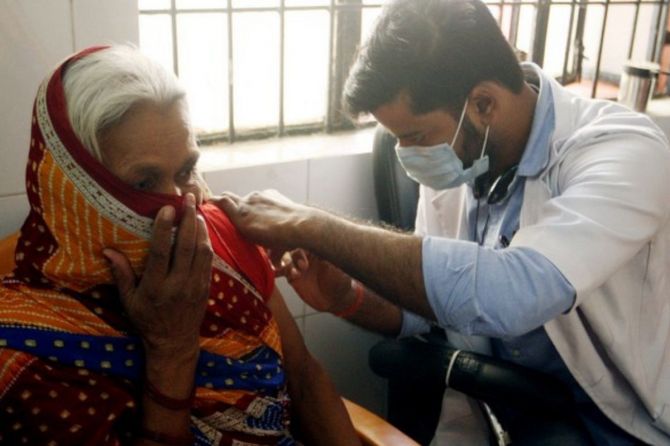
In an exclusive interview with ANI, RS Sharma said, "The elderly population that comes above 60 years of age with comorbidities is eligible for COVID-19 'precautionary doses', will have to take a medical certificate to take the dose. The other process of vaccination will remain the same as before. The CoWIN application has all the details. They have already got two doses, they can go with their Comorbidities certificate and then get the third dose."
Dr Sharma added that the medical certificate of comorbidity should be signed by any registered medical practitioner that can be uploaded and even beneficiaries can take that along as a hard copy to the vaccination centre.
Explaining further, he said, "There is already a detail about comorbidity certificate which was published initially when we were doing comorbidity for 45 to 60+ age group. The same formula will apply now also the same certificate will be required for the same comorbidities condition."
There are twenty medical comorbidities criteria including diabetes, kidney disease or on dialysis, cardiovascular diseases, stemcell transplant, cancer, cirrhosis, sickle cell disease, current prolonged use of steroids or immunosuppressant drugs, muscular dystrophy/acid attack with involvement of respiratory system/persons with disabilities having high support needs/multiple disabilities including deaf-blindness and severe respiratory disease with hospitalisations in last two years.
A booster dose certificate will also be given to beneficiaries.
Earlier on Saturday, Prime Minister Narendra Modi in his address to the nation, had said that the precautionary doses of vaccine will be started for healthcare, frontline workers and citizens above 60 years with co-morbidities starting January 10, 2022.